Truth is the property of no individual but is the treasure of all men. Ralph Waldo Emerson
A label from the collection of Archduke Stephan, dating in the mid 1800s
I often get asked why I collect minerals, and in general I ignore the inquiry because the answer is a thesis not a sentence. Recently I had returned to me several silver specimens from my collection that “disappeared” for 2 years. The conditions of the “disappearance” is a tale of poor decisions (mine), disorganization (a middle man) and opportunistic dishonesty (a mineral dealer of questionable ethics). However, fate and friends dealt a favorable hand and the specimens were returned (although one was damaged), and my joy in return of the prodigal stones gave me a chance to explain my rationale for collecting.
Frieberg silver wire, 6 cm high. I acquired this specimen in 2010, and it was first cataloged in a collection in 1832.
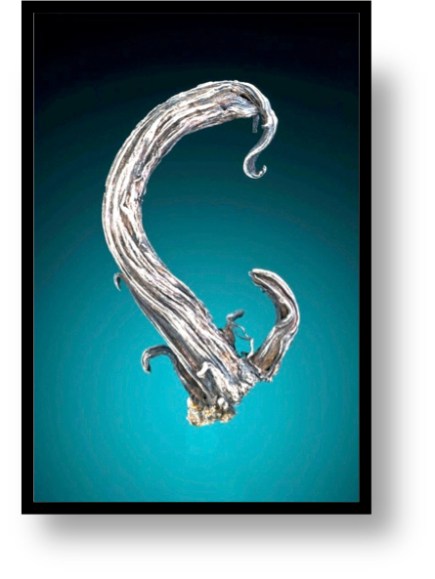
The centerpiece of the missing specimens was a silver wire from the great German locality of Freiberg. The specimen is a little over 6 cm high and has a patina of age giving it a glow of significance. The specimen was first documented to be in a collection in 1832, and it passed through at least 9 different owners before it came to me. The specimen has beauty to me, but more importantly, it is an artifact of history and humanity. This particular specimen has aesthetics, and its form is an interesting mineralogical tale. In addition it is from a mining locality that has a rich history, and once the silver wire was mined, it was a prized natural history specimen that was passed along to collectors that had the same passion as I.
Collectors: Evolution or Illness
There are dedicated collectors in every society, and these collectors are not defined by economic or social class. There is a large body of literature on the psychology of collecting (most of which I find pompous and over reaching!), and there are two basic schools of thought. The first is the Freudian view that says collectors are afflicted with a compulsive disorder; collecting is emotional and a desire to control or connect. The second view is the collecting is an evolutionary trait associated with amassing treasure as a survival instinct. Neither of these synopses really describes the passion that most serious mineral collectors I knew feel.
The vast majority of mineral collectors I associate with feel joy in finding a natural object that has beauty and form. There are mineral collectors that pursue specimens as investment or status. However, they are usually of the “moneyed class”, and they represent something different that most of the collectors I know, although the first prominent mineral collectors were indeed from the rich and powerful. Mineral collecting began in the 18th century by aristocrats – they assembled cabinets of rocks and minerals, and these cabinets were badges of social class. Perhaps the most famous of these early aristocratic collectors was Archduke Stephan Franz Victor von Habsburg-Lothringen. Born into the Hapsburg Royal Court, Stephan was well educated, and destined to a life not sullied by common labors. He built a mineral collection and cabinet that eventually contained more than 20,000 specimens. The top figure in this posting is one of Stephan’s labels – there are many mineral collectors that value a Stephan label almost as much as a mineral. The stephanite specimen below is also from Freiberg, and was once in the Archduke’s collection. Stephanite is named for Stephan.
Frieberg stephanite, 7 cm high, acquired in 2008. This is an extraordinary stephanite crystal group, and has spent part of its “life” in two museums and three private collections before coming to my collection.
Collecting Silver
I first started collecting minerals at age 4 or 5, fostered by the passion of my father who loved collecting minerals in the field. At least a couple of times a month we would journey to mines or mineral localities in New Mexico, Colorado or Arizona. I can’t really say why my father was such a dedicated field collector – he was a chemist by profession, but the science of minerals did not seem to be what was important to him. He was raised in the home of his grandfather who was a prospector in Arizona, and this man seemed certain that the next great lode was hidden in the deserts and rugged mountains of Arizona just waiting to be discovered. This lust of treasure hunting more describes my father’s passion – he was not really looking for the mother lode, but he loved finding a great specimen in the ground. Once we got the rocks home he was far less interested in them – the pursuit was his passion. He built an extraordinary library for topographical mineralogy – boxes and filing cabinets filled with Xeroxed reports and papers from obscure journals. He assembled this material to map out where to go and collect next.
My brothers and sisters often accompanied my father on our journeys through the southwest. However, none of them became mineral collectors, nor even really dabbled in collecting. Clearly, mineral collecting is not a simple matter of nurture. My first mineral collections were mostly driven by form – I loved euhedral crystals with sharp faces. By age 10 I had a catalog for my collection that numbered in the several hundred; within a few years after that I was actively trading many of my specimens with a dealer in Albuquerque in an attempt to acquire “better” material. In high school I had my first serious cull of my collection after which I would only collect sulfide ore minerals. I had a very fine collection of galena, pyrite, chalcopyrite and a few chalcocites!
Freiberg acanthite, 5.5 cm high. This specimen was acquired in 2002, and has been pictured in numerous publications
I continued to refine my collecting until the early 1980s when I decided to only collected silver minerals. Although I am interested in nearly all minerals, my focus is quite narrow. There about 4600 different mineral species known, and approximately 160 of them have silver as an essential element; of these, only about 15 are “common” or available as crystals that are easily seen with the naked eye. Silver has been reported from more than 20,000 localities world wide – approximately 100 different localities have produced quantities of very well crystallized specimens of the common silver species. In my collection today I have samples of 109 of the different silver species, and I have every important locality represented.
Freiberg
The wire silver from Freiberg is a quintessential specimen from my collection. The Erzgebirge are a modest mountain range that runs along the boundary between southeastern Germany and the northern part of the Czech Republic) for about 100 km. The English translation of Erzgebirge is “Ore Mountains”, and these rolling hills are the birth place of modern mining, metallurgy and mineralogy. The German side of the Erzgebirge is known as the Saxony side, while the Czech side is referred to as Bohemia. The Bohemia mines of fame include Kutna Hora and Jachymos/St. Joachimsthal, while the Saxon mining areas of note are Schneeberg and Schlema, Annaberg, Marienberg, Johanngeorgenstadt, and the most famous of all, Freiberg.
The story of the Erzgebirge silver is voluminous topic; a simple summary of Freiberg serves to at least stake the claim of the Ore Mountains as being the most important silver mining camps in history. Silver was first discovered in Freiberg in 1163 – the area is located about 30 km west-southwest of Desden. The town was officially founded in 1186, and over 800 years of mining produced about 8 kilotonnes of silver. The two most famous Freiberg mines are the Himmelfahrt and Himmelsfurst – these were large mines with multiple shafts. The enduring influence of Frieberg came with the founding of the Bergakademie Frieberg, or Frieberg Mining Academy, by Prince Franz Xaver in 1765. The mining academy in Freiberg can now lay claim to the oldest School of Mines, and can lay claim to educating some of the most famous mining engineers and mineralogist in the world. A.G. Werner, a mining geologist on the faculty first proposed a chemical classification of minerals in 1774 – he invented the modern scheme for describing minerals. The Frieberg Academy had a profound effect on mineralogy also be preserving specimens that came from the mines and build a remarkable mineral collection.
Freiberg silver, 7 cm high, acquired in 2001. This is a classic example of wire silver that must have grown from the decomposition of acanthite. The wires have been exposed by removing the encasing calcite.
I had the chance to visit the Freiberg Academy in the summer of 1991. The Berlin wall had just fallen, and East and West Germany had reunified. It was clear as I drove from Frankfurt to Dresden that there really were two Germanys. The infrastructure in the east was third world, and as I drove through Dresden there still were Soviet tanks deployed. However, when I got to Freiberg, the Academy staff were incredibly warm and helpful. What I saw in the collections was amazing, and made my connection to my Freiberg silver minerals much richer.
Freiberg Pyrargyrite, field of view is 1.7 cm. This specimen was acquired in 1984, a came through a dealer that had traded it out of the American Museum of Natural History in New York. The AMNH is one of the great mineral museums in the world, and received the collections from the Columbia School of Mines in the early part of the 20th Century.
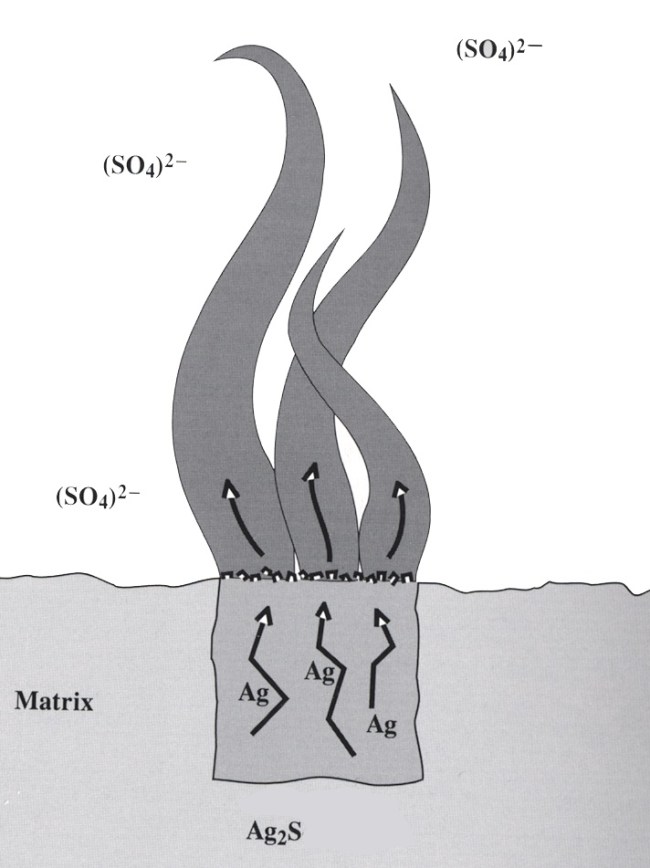
Silver has an affinity for anions of sulfur, selenium and tellurium, all of which have similar ionic radii. These minerals are known as the silver sulfides (in the nomenclature of Dana, these include the tellurides and selenides), of which the acanthite group is the most common. The acanthite group includes the simplest sulfides (the most common of these are acanthite, argentite, aguilarite, naumannite, hessite, petzite, empressite, jalpaite, stromeyerite and eucairite). This group of minerals displays the characteristic of temperature-dependent dimorphism. At high temperatures these minerals are usually cubic or hexagonal, whereas at lower temperatures the minerals display an orthorhombic or monoclinic crystal structure. The transition temperature is usually between 130 and 180o C. Acanthite and argentite are the most common dimorphic pair. Acanthtite is the form that is stable at room temperature, so even when a specimen appears to have cubic crystals, it is a monoclinic microstructure frozen in the cubic frame. The same thing that makes the silver-sulfur bond temperature dependent also makes acanthite sensitive to decomposition when temperature and pressure change – silver is released from the sulfur bond and grows wires out of the acanthite. Silver wires are extremely common, and it is clear that they are all formed by the decomposition of a silver sulfide (most likely acanthite). This was first observed and understood at Freiberg.
An early explanation for the growth of silver wires by the decomposition of acanthite
The rest of the story of the mineral mystery
I have wanted to write a book on the silver minerals for a long time. Gloria Staebler has provided me the encouragement to pursue this book which will be years in the making. Along the way I decided to illustrate certain aspects of the mineralogy with pictures of many of my specimens. Photographing minerals specimens is not easy under the best of circumstances, and silver minerals are extremely difficult. Their dark color, intergrown crystals, and occasional high luster means that most attempts to capture their beauty with a camera result in images that closely resemble black ink blots. With this in mind, I sent a subcollection of the specimens to be photographed by one of the best mineral artists in the world. However, sending multiple specimens to be photographed far from my immediate control was a poor decision. It took several attempts to get the images right; over time one small box of the specimens are returned to the wrong owner. Although I did not get back my specimens there was no documentation that I did not get back these back – in fact, many people assumed I simply had misplaced them. I knew that was not the case, but I was frustrated in locating the silvers.
Advertisement in the Mineralogical Record that featured my Freiberg silver. Read the ad – this is what is wrong with mineral collecting today.
In late February of this year I received the March-April issue of Mineralogical Record. As usual, I first read the most interesting article to me, and then thumbed through the rest of the volume looking at advertisements from various mineral dealers. As I turned the pages I was stunned – there was a picture of my Freiberg silver in the ad of dealer for sale. I was outraged! Indeed my specimens had been returned to the wrong owner, but that dealer chose to assume that mistake was fortuitous! Found wealth! The repatriation was emotional and messy, but I am reminded again that honesty is a rarer commodity than it should be. The fortuitous dealer claims that he did nothing wrong at all – in fact, he simply just thought the minerals were his, and he had forgotten how he got them.
This story is not done, but in most ways the universe is again right. However, the story of a mineral lost is also a tale about collectors and the mineral hobby. When I first started in the hobby more than 50 years ago it was different. There were far more scholars than today. My sense is that this is not because there is less interest in mineralogy, but because the opportunities to build a meaningful collection are greatly diminished. Prices have escalated – this is always true in collectables – but in a very dramatic way collecting is out of reach for the person of average means. I have benefited occasionally from this “art pricing model”; specimens I bought for hundreds of dollars I have traded or sold for 100 times purchase value. New collectors do not have the “hundreds of dollars” specimens available to trade or sell to better create a collection, and thus, they tend to drift away. The case of the dealer wanting to sell my “fortuitously” purloined silvers is symptomatic of the commercial side of the collecting equation. Not something to be happy about, nor do I see an enlightening horizon.