The universe is built on a plan the profound symmetry of which is somehow present in the inner structure of our intellect, Paul Valery, 19th century French Poet

Silver, Kearsarge Mine, Houghton Co., Michigan. The specimen is 3.5 cm across. This native silver is a lustrous example of a “weave” of sliver crystals exhibiting spinel twinning. Photograph by Jeff Scovil. Click on any photo in the article to get a larger view.
When I first started building a mineral collection — back about 1960 — the single most compelling criteria for determining if a specimen was a “keeper” or just something for the beer flats filled with colorful, yet, unworthy rocks, was whether there was a euhedral crystal. My fascination with the perfection of a sharp crystal face is not at all uncommon for beginning collectors. The fact that nature could take time to construct something so perfect strikes a deep chord; the vast universe created by the ultimate act of violence – the big bang – and ruled by entropy, and inevitable decay, still values symmetry. I recall an early discussion with my mother on the beauty of spring flowers – I asked her why she thought they were beautiful, and she responded with a joyful exposition on the bright and varied colors and the delicate nature of the pistil, and remarkable symmetry of the petals. I told her that the petals were exactly like crystals since they are always alike, and must be following some sort of “rules”.
The English word symmetry comes from the Greek symmetria; in turn, symmetria is a concatenation of Greek words sun and metron, meaning “together” and “measure”. There is a substantial body of Greek literature that refers to symmetry as “harmonious and beautiful proportion and balance”. This philosophical definition of symmetry deviates from the strictly mathematical definition, but still projects the power of something that is predictable and has a geometric balance to be pleasing to the eye. This “pleasing to the eye” is a euphemism for beauty — hard to define exactly, but beauty excites our aesthetic senses.
To me, there is nothing more pleasing to the eye than a silver specimen exhibiting spinel twinning – repeating patterns of crystals that produces a highly geometric weave. The photograph at the top of the column is a silver from the Kearsarge Mine, Houghton Co., in the Upper Peninsula of Michigan. The specimen is defined by a central rib — an elongated stack of silver octahedrons, and branches intersecting the this rib at angles of approximately 60 degrees. In turn, these branches have secondary branches exiting at similar angles. The repeating geometry yields a specimen architecture that is clean and sharp – an exemplar of what the Greeks meant by symmetry.

Silver, from a locality near Tongchong, Yunnan Province, China. Specimen height is 6cm. The wires are the result of decomposition of acanthite. Jeff Scovil photograph.
Silver: a special element
Silver is a remarkable element that can form an array of minerals; about 180 different species. Thankfully, elemental silver is sufficiently inert to occur in nature and is widely distributed throughout the world. Native silver is a metal of bright white color; it has the highest reflectivity of any metal. Silver is also the best metallic conductor of heat and electricity and extremely malleable and ductile. These properties are, of course, a result of crystal and atomic structure, which is a face-centered cube with metallic bonds. The atomic radius of silver is nearly identical to that of gold – and gold commonly substitutes into the silver crystal lattice.

Native silver crystal forms, from Goldschmidt (1913). The octahedron is most common, although cubes are much coveted by collectors. The bottom 3 figures show spinel twinning.
Silver crystallizes in the isometric system, and although individual, sharp macro crystals are rare, the octahedron and cube forms are most common. The largest individual crystals are from Kongsberg, Norway, where some octahedrons 3-5 cm on a side grace a few fortunate collections.

Cubic silver in calcite; Kongsberg, Norway.Each of the large cubes are approximately 1 cm on a side. Jeff Scovil photograph.
As a rule, crystals of silver are equi-dimensional or platy. The platy nature comes from the propensity from silver to twin (this is similarly seen in gold and copper) on octahedral faces {111}. This twinning is known as spinel twinning and is described below. The conditions for when spinel twinning occurs appears limited – although silver masses of spinel twins are known from hundreds of locations, there are only three or four localities where this type of silver crystallization is common. The two most famous localities are Batopilas, Mexico and Chanarcillo, Chile.
By far, the most common form of silver in a mineral collections is the wire – which is a secondary growth from the decomposition of silver sulfides and sulfosalts. Any silver deposit that undergoes supergene enrichment inevitably has silver wire specimens. The picture at the top of this section of the article is a fine silver wire mass from Tongchong in China. The wires in this specimen are attached to a very small piece of acanthite, no doubt the host material that provided the silver. All silver sulfides and most silver sulfosalts will produce silver wire upon disassociation — especially promoted by heating. Although wires can be extremely interesting and coveted specimens for collectors, there are been numerous cases where the wires were “grown” by unscrupulous collector/dealers and passed off as “natural” specimens.
Beauty in Nature
When I hear the word “beautiful” used to describe minerals by collectors I often ask what they mean. More often than not, the answers seem wanting to me. Mostly, it is about color — pink and red minerals are always “beautiful”, but black and brown minerals are “interesting”. Although color can transfix, and certainly evoke emotion, I can not relate to it as the primary metric of natural beauty. I am also looking for structure in my surroundings – a window into the soul of nature, order out of the chaos all around me.
A seminal event for me was attending “math summer camp” during the summer between 7th and 8th grade. The instructor was an outstanding teacher named Jack Gehre, and his focus was geometry and trigonometry. Early in the class Mr. Gehre introduced Euler’s formula; for any normal polyhedron, the sum of the number of faces plus the number of vertices, minus the number of edges always equals 2. I spent the rest of the summer camp trying to understand why. I suddenly had a “rule” in nature that I could apply to my mineral collection — a rule mysterious and powerful, but incredibly simple. It was beautiful. I did not know much about Euler then, but later in college I was introduced to another “law” by Leonhard Euler — an incredible 18th century Swiss mathematician — that has to be the most beautiful equation in all of nature. I was in a class on series analysis, and the professor, Alan Sharples, walked in the first day of the semester and wrote the following on the black board:
Sharples said, “this is Euler’s identity, a remarkable assertion. Prove it. That is all for this first day of class.” Turns out that this is pretty easy to prove, but when I viewed this on the blackboard I was transfixed — it was pure beauty. Imagine three essential mathematical constants – e, pi, and i – combined to equal -1. Wow – simple, brief, and exact. To this day I view this as a definition of beauty (Euler’s identity is routinely identified as the one of the most beautiful equations in science).
Euler’s identify may seem a long ways from realm of beauty in the mineral kingdom. However, to me, they are very much related. Simple, surprising, and an expression of natural symmetry.

Herringbone silver mass, Batopilas, Chihuahua, Mexico. The Specimen is 3.75 cm across. Jeff Scovil, photograph.
Respect the Spinel Twin
Crystal growth in nature is quite complex; the crystal form, crystal size, crystal chemistry all are expressions of the paragenesis. Crystallization for most geologic materials involves the precipitation of a solid (the crystal) out of a solution or solvent (usually hot thermal fluids, although solutions of nearly any temperature can carry dissolved loads of ions and cations). Crystals start with nucleation of a few molecules from the solution, and then growth occurs by pulling the necessary ionic components out of solution. The rate at which individual crystals grows depends strongly on the saturation level of the ions of interest – supersaturated solutions appear to be able to grow crystals at extraordinary rates (at least compared to geologic time!), sometimes at several cubic cm per hour.
It is not clear who first recognized twinning in crystals, but it was first written about in detail by Rene-Just Haüy in his epic tome Traité de Minéralogie, published in 1801. In the beginning part of the 20th century there were a number of studies to understanding twinning in minerals. The classic definition was introduced by Friedel in 1926: A twin is a complex crystalline edifice built up of two or more homogeneous portions of the same crystal species in contact (juxtaposition) and orientated with respect to each other according to well-defined laws. The “well-defined laws” all are based on some simple ideas, the most important of which is that within a crystal core that a least one lattice row (i.e., a crystal edge) is common to two different crystals. The figures below illustrate this concept — the lattice of a cubic crystal is defined by four points, and a plane can be drawn through these points that allows a second crystal to share lattice points but have a rotated orientation. Twinning adds symmetry to a crystal aggregate, most commonly about a rotation axis or reflection across a plane. In the metals copper, gold and silver, a particular type of twin is common, called the spinel twin.
Spinel twins are so-named because it is a very common habit seen in the mineral spinel. They are contact twins, meaning that have a planar composition surface shared by two individual crystals; this surface is along an octahedral face (written as {111}), and means that there is a rotation of 180o about the contact plane. This is illustrated by the lower figure above – there are two octahedrons joined along a contact plane, but the top terminations “point” in directions and are separated by 120o. The figure below shows how spinel twins can be flattened, and give the characteristic triangular faces that are seen on platy crystals of silver (and gold).
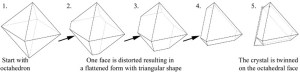
Notional relationship between an octahedron and a spinel twin producing a triangular type crystal face. This is “notional” in that this is NOT how the spinel twin evolves with time, but rather, a visual guide to compare an octahedron (left) and triangular face (right).
In silver, spinel twinning almost always repeats itself with regularity, producing a pattern that resembles a weave of wires. The silver at the top of this section of the article is from Batopilas, Chihuahua, Mexico, and is an example of a mass of spinel twins. Through the middle of the specimen is a series of parallel elongated crystals, and growing “off” these strands are regular strands oriented at 60 degrees (or 120 degrees, strictly speaking). These are all spinel twins – repeating some natural frequency that is due to a long lost geologic condition. Once assembled, the spinel twins from an aggregate of crystals that has been called a “herringbone” silver in reference to the similarity to the shape of the rib cage of the smelly game fish beloved by the peoples of the Baltic.
Why do spinel twins form in silver? Under certain ideal conditions, a single large crystal represents a “minimum” energy condition, and thus is due to an important thermodynamic rule — a chemical system will stabilize at state of least energy. If individual crystals are a minimum energy state, then twinned crystals are by necessity at state of higher energy, and thus should be rare. However, environmental conditions tend to localize energy states; for a supersaturated solution, the crystal growth is extremely rapid, and twinned crystal allow more ions to join a crystallize aggregate faster, thus minimizing a local energy state. For all “herringbone” silver specimens it appears that the conditions of formation require a supersaturated solution, low in concentrations of sulfur, and extremely rapid crystal growth. These conditions are relatively rare in most epithermal vein deposits; it is very uncommon to find a spinel twinned silver specimen from the great silver deposits of Colorado, Ontario or Freiberg!

Large silver plate (7.5 cm from side-to-side) from Chanarcillo, Chile. The specimen is a weave of spinel twins – and is my favorite silver specimen in my collection. Jeff Scovil photograph.
The silver pictured above is my favorite native silver in my entire mineral collection. This is a large “herringbone” plate with a three dimensional repeating pattern of twins. The specimen represents something remarkable in turns of crystal growth. The tiniest variations in chemistry or temperature during growth would have truncated the growth of this silver weave.

Close up of the Chanarcillo specimen shown above. The central rib is an elongated chain of octahedrons. Field of view is 3 cm. Repeatedly, silver crystal “twin” off the octahedral face(s). At the very top of the specimen there are identifiable octahedrons.
A close examination of the Chanarcillo herringbone yields views of spectacular detail – endlessly repeating, and shouting the fundamental rules for symmetry in crystals. Along the edges of the crystalline mass you can see individual octahedrons – the termination of various elongated crystals.
Beauty and the pretenders
Rapid growth in silver often produces crystalline masses that are complex. However, spinel twins are distinct, and uncommon. Rapid growth often leads to dendritic masses – mostly silver feather patterns or strings of stacked cubes. These dendrites are not spinel twins; in fact, instead of fundamental order, they represent chaotic growth. Although there is some sense of beauty in the randomness of dendrites, it is mostly through “self-similarity” – various patterns that appear to scale with size. This is fundamentally different than ordered spinel twins – and in many ways points to disordered processes. I am always shocked (okay, probably an overly harsh expression of emotion) when I find dealers selling “herringbone” silvers that are in fact dendrites. That is like marketing hamburger as Filet Mignon. Similarly, silver wires can certainly be attractive; however, they are products of mineral destruction not construction. To me, beauty in silver spinel twins is about construction, order, and symmetry. Defining beauty will allows be in the eye of the beholder — it is just better when there are rules involved.
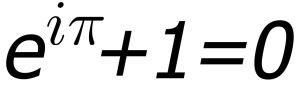


Dear Sir,
I have read the articles in your blog, and have enjoyed it.
I want to send some samples of native silver I have collected as tokens of appreciation.
Please can you tell me your address, to send them.
best regards.
yours faithfully,
Joan Abella i Creus.
http://www.mineralogicalrecord.com/labels.asp?colid=1358
http://mineralsabella.blogspot.com.es/search/label/PLATA%20ANTROPOG%C3%89NICA%20%3F